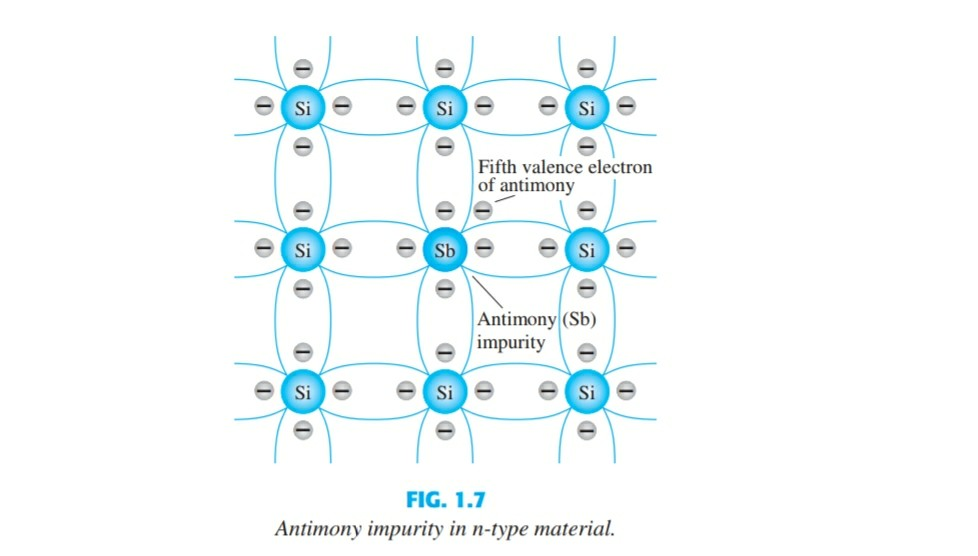
Mårtensson, "Core-Level Binding Energies in Metals," J. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Here Germanium has 4 valence electrons whereas Antimony belonging to group 15 has 5 valence electrons and hence when these are dopped one electron remains. 
Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. To write the electron configuration of antimony, we should first know the total number of electrons present in an. Nitrogen, phosphorus, and arsenic can form ionic compounds by gaining three electrons, forming the nitride (N 3- ), phosphide (P 3-) and arsenide (As 3-) anions, but they more frequently form compounds through covalent bonding. 
They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The Group 5A elements have five valence electrons in their highest-energy orbitals ( ns2np3 ). Antimony has the chemical symbol Sb and is considered a metalloid. Take the look of Antimony valence electrons with our Lewis dot diagram. Antimony has 51 protons, 51 electrons, and 71 neutrons. The element increases the hardness potency of alloys to make it stronger. The data are adapted from references 1-3. Further, the rest of Antimony is useful to use as the pure metal element with the alloys. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. Connect each atom to the central atom using single bonds first. If these atoms are not present, then go with the atom you have least of in the formula. Carbon is always central and Hydrogen is a terminal atom. The n-type semiconductor is created by introducing those impurity elements that have five valence electrons (pentavalent), such as antimony, arsenic. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. Remember valence electrons are the roman numerals above the element. All values of electron binding energies are given in eV. Antimony (Sb, 51) has 5 valence electrons (and so do all the other elements in that group.) What company has 5 valence electrons Group 15 elements (N, P, As, Sb, Bi) What element has. 1967, 47, 1300.Įlectron binding energies Electron binding energies for antimony. These effective nuclear charges, Z eff, are adapted from the following references: Effective nuclear charges for antimony 1s
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
